Tapirus haysii
Quick Facts
Common Names: Hays’ tapir, giant tapir
Tapirus haysii is a moderately large species of tapir, larger than the modern lowland or Brazilian tapir (Tapirus terrestris) and similar in size to Baird’s tapir (Tapirus bairdii).
Tapirus haysii is part of an extinct lineage, with no living descendants.
Analysis of the tooth enamel indicates a browsing diet of leaves from bushes, shrubs, and trees, as is typical of the genus.
Age Range
- Early Pleistocene Epoch; late Blancan to early Irvingtonian land mammal age
- About 2.5 to 1.0 million years ago
Scientific Name and Classification
Tapirus haysii Leidy, 1859
Source of Species Name: Named for Dr. Isaac Hays (1796-1879), an ophthalmologist in Philadelphia who was interested in natural history, especially paleontology. He obtained the type specimen of the species from its collector Thomas Nuttall and donated it to the Academy of Natural Sciences of Philadelphia in 1852 (Ray and Sanders, 1984).
Classification: Mammalia, Eutheria, Laurasiatheria, Perissodactyla, Tapiromorpha, Ceratomorpha, Tapiridae
Alternate Scientific Name: Tapirus copei
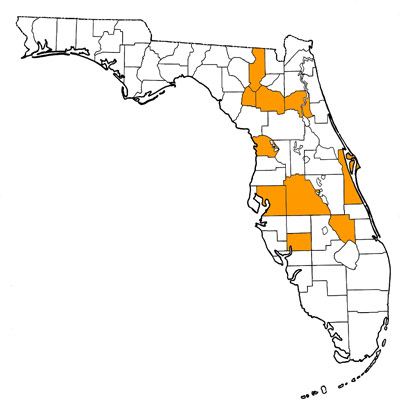
Overall Geographic Range
Eastern, central and mid-western United States, including occurrences in Colorado, Nebraska, Oklahoma, Texas, Pennsylvania, South Carolina, and North Carolina. Type locality is Benners Estate, Neuse River near New Bern, Pamlico County, North Carolina (Ray and Sanders, 1984).
Florida Fossil Occurrences
Florida fossil sites with Tapirus haysii:
- Alachua County—Haile 16A; Haile 21A; Pareners Branch
- Brevard County—Tucker Borrow Pit
- Citrus County—Inglis 1C; Inglis 1D
- Columbia County—Santa Fe River 1
- De Soto County—De Soto Shell Pit 5
- Gilchrist County—SR138 Spring Site
- Hillsborough County—Apollo Beach; Caloosa Shell Pit; Leisey Shell Pit 1A; Leisey Shell Pit 1B; Leisey Shell Pit 2; Leisey Shell Pit 3; Leisey Shell Pit 3A; Leisey Shell Pit 3B
- Okeechobee County—Kissimmee River
- Polk County—Fort Green Mine; Fort Meade Mine; Palmetto Mine; Payne Creek Mine; Phosphoria Mine; Pool Branch Site , Peace River Mine
- Putnam County—Devil’s Elbow 2
Discussion
Tapirus haysii is a moderately large species of tapir, larger than the modern lowland or Brazilian tapir (Tapirus terrestris) and similar in size to Baird’s tapir (Tapirus bairdii) which today lives in southern Mexico, Central America, and northwestern South America. Among other species in the genus Tapirus, Tapirus haysii is most closely related to the other Pleistocene species from the eastern United States, Tapirus veroensis and Tapirus lundeliusi. Together they form the subgenus Helicotapirus (Hulbert, 2010). Helicotapirus is an extinct lineage, with no living descendants.


Simpson (1945) declared that Tapirus haysii was an invalid scientific name and proposed the name Tapirus copei to replace it. This action was followed for about 40 years (e.g., Ray, 1964; Kurtén and Anderson, 1980). Ray and Sanders (1984) demonstrated the validity of Tapirus haysii and definitively showed that its type locality is from North Carolina (prior to this there was considerable confusion in the scientific literature about the true type locality of this species). Since 1984, most researchers have followed the conclusions of Ray and Sanders and accepted Tapirus haysii as the valid scientific name for this species, andTapirus copei has been considered its junior synonym.
Tapirus haysii differs from the common middle to late Pleistocene tapir from the eastern United States, Tapirus veroensis, by its relatively longer post-canine diastema, sharply downturned lateral edges of the nasal bone, and, on average, much greater size. In other features of the skull, the two are identical. The size difference is more pronounced for the skull and teeth and less so for the limb bones. Evidently the head of Tapirus haysii was relatively large compared to its body. Small individuals of Tapirus haysii overlap in size with large individuals of Tapirus veroensis. At least in Florida, the two species do not co-occur; all well dated records of Tapirus haysii are older than those of Tapirus veroensis. Note that in the early 1900s, relatively large individuals of Tapirus veroensis were often identified in the scientific literature as Tapirus haysii.

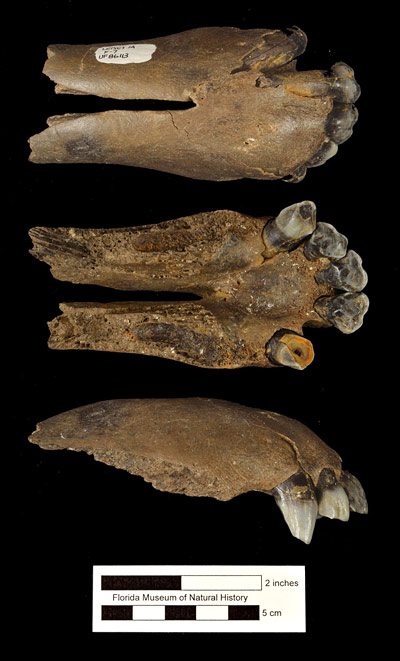
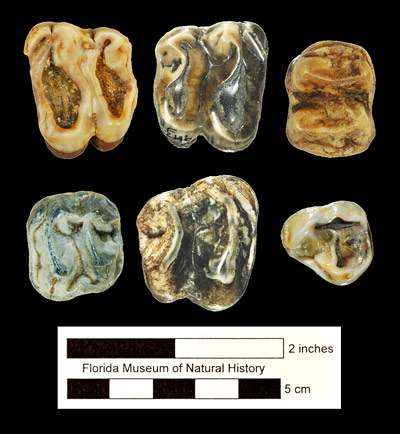
The best samples of Tapirus haysii are from Port Kennedy Cave (Pennsylvania) and Leisey Shell Pit (Florida). Analysis of the relative proportions of stable carbon isotopes from tooth enamel taken from Leisey 1A specimens by DeSantis et al. (2009) indicated a browsing diet of leaves from bushes, shrubs, and trees, as is typical of the genus.
Supposed differences in the first and second upper premolars between Tapirus veroensis and Tapirus haysii that were stressed by Simpson (1945) have not been borne out as sample sizes and the known variation in premolar anatomy have increased. Another factor complicating identification, particularly of isolated teeth, is that the first molars of Tapirus haysii are about the same size as the second molars of Tapirus veroensis. This means that proper identification of tooth position is a critical first step in correctly identifying isolated teeth and jaw fragments.
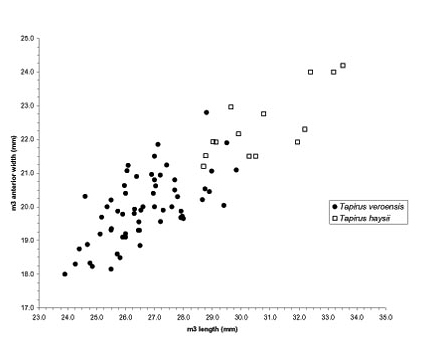
In Figure 7, lower third molar length is plotted on the X axis and lower third molar anterior width is plotted on the Y axis. Specimens with lengths less than 28.7 mm or widths less than 21.2 mm should be identified as Tapirus veroensis; those with lengths greater than 30 mm or widths greater than 23 mm should be identified as Tapirus haysii. Specimens in the zone of overlap are indeterminate, although if they are heavily worn they are most likely Tapirus haysii, but if unworn or very slight worn, then most likely Tapirus veroensis.
The chart below will allow identification of most isolated tapir teeth from the Pleistocene of Florida and the rest of the eastern U.S. Specimens of intermediate size cannot be assigned with confidence to a single species without other evidence. In the table, L = length in millimeters; W = anterior width in millimeters; PW = posterior width; P = upper premolar; p= lower premolar; M = upper molar; and m = lower molar.
| Tooth position | assign to Tapirus haysii | assign to Tapirus veroensis |
| P1 | L > 20.6 or PW > 15.0 | L < 19.7 or PW < 14.7 |
| P2 | L > 22.0 or W > 23.3 | L < 20.6 or W < 22.0 |
| P3 | L > 22.6 or W > 26.6 | L < 21.6 or W < 25.7 |
| P4 | L > 23.6 or W > 29.3 | L < 22.6 or W < 27.0 |
| M1 | L > 25.5 or W > 28.8 | L < 23.9 or W < 26.9 |
| M2 | L > 27.6 or W > 31.9 | L < 25.5 or W < 30.8 |
| M3 | L > 27.9 or W > 31.7 | L < 27.0 or W < 31.5 |
| p2 | L > 25.5 or PW > 17.3 | L < 25.5 or PW < 15.4 |
| p3 | L > 24.6 or W > 17.4 | L < 22.5 or W < 16.8 |
| p4 | L > 24.5 or W > 21.2 | L < 22.5 or W < 19.9 |
| m1 | L > 25.8 or W > 20.8 | L < 23.0 or W < 19.9 |
| m2 | L > 27.6 or W > 22.3 | L < 27.0 or W < 21.8 |
| m3 | L > 29.8 or W > 22.8 | L < 28.7 or W < 21.2 |
Sources
- Original Author(s): Richard C. Hulbert Jr.
- Original Completion Date: February 26, 2009
- Editor(s) Name(s): Richard C. Hulbert Jr. and Natali Valdes
- Last Updated On: March 3, 2015
This material is based upon work supported by the National Science Foundation under Grant Number CSBR 1203222, Jonathan Bloch, Principal Investigator. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation.
Copyright © Florida Museum of Natural History, University of Florida